National Hemophilia Foundation (NHF) - Posters
Non-severe hemophilia is not benign? - Insights from the PROBE Study |
|
|
|
|
Objective:
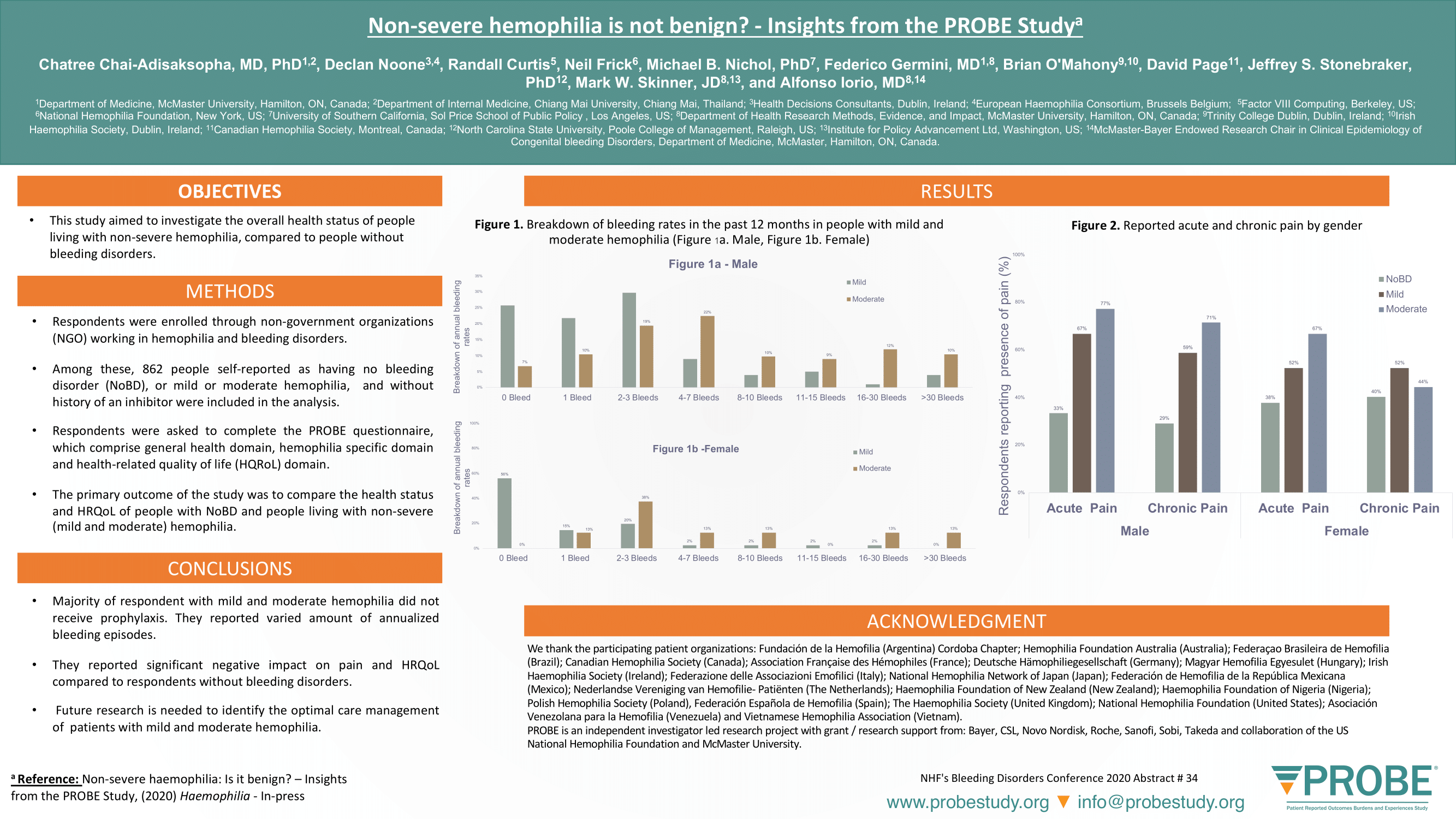
There are limited data on the impact of haemophilia on health status and health-related quality of life (HRQL) in people affected by non-severe haemophilia. Aim is to evaluate the health status of people living with mild or moderate haemophilia.
Methods:
A cross-sectional, multinational study was conducted as part of the Patient ReportedOutcomes, Burdens and Experiences (PROBE) project. Respondents without bleeding disorder(NoBD) and those with mild or moderate haemophilia were included. Respondents were askedto complete the PROBE questionnaire, which contains haemophilia-related questions, generalhealth questions and HRQL. Results were compared between unaffected individuals and peoplewith mild or moderate haemophilia.
Summary:
A total of 862 respondents, of whom 144 with moderate haemophilia, 143 with mildhaemophilia and 575 with NoBD were included. Median age (first-third quartile) was 33 (23-46),42 (25-55) and 43 (35-54), respectively. In relation to bleeding in the previous 12 months,respondents with mildreported less bleeding frequency than those with moderate haemophilia, with similar patterns of bleeding frequency seen in both male and female cohorts. Reporting of acute pain is less in those with NoBD compared to the mild to moderate cohorts for both genders (male - 33%, 67%, 77%; female - 38%, 52%, 67%, respectively). Thirteen percent of those with NoBD reported an impact on activities of daily living compared with mild andmoderate haemophilia who reported of 35% and 61%, respectively. The impact on quality of life due to mild haemophilia compared to those with NoBD was areduction of 5.2%, 5.0% and 9.3% in VAS, EQ-5D-5L and PROBE Score respectively (p?0.001).
Conclusions:
People affected by mild or moderate hemophilia encountered a significantamount of haemophilia related sequalae. Future research is needed to identify the optimalmanagement of moderate and mild hemophilia patients, with particular focus on earlyidentification of patients with a severe clinical phenotype.



